Brain tumors
Treatment Specialization with CLRD’s Integrated Diagnosis, Precision Surgery, and Regeneration Pathways
Brain tumors, whether primary gliomas, meningiomas, pituitary adenomas, or metastatic lesions, pose a dual challenge: they threaten life by invasive growth and raised intracranial pressure while risking long‑term disability by encroaching on eloquent networks governing movement, speech, memory, vision, and executive function. The therapeutic mandate is therefore uncompromisingly twofold: achieve the maximal safe resection to control disease biology and, at the same time, preserve or recover cognition and motor performance. This article presents a full-spectrum, treatment‑focused overview of how that mandate is met in contemporary neuro‑oncology and details how CLRD’s capabilities, shaped by decades of translational expertise in cellular therapies, organ bioengineering, and nanotechnology are applied in the diagnosis and treatment of brain tumors. While this page concentrates on treatment specialization rather than research reporting, it is informed by the breadth of CLRD’s published work across neural regeneration, stem cell biology, organ scaffolding, and precision biomarker science.
Understanding the Functional Stakes and Therapeutic Objectives
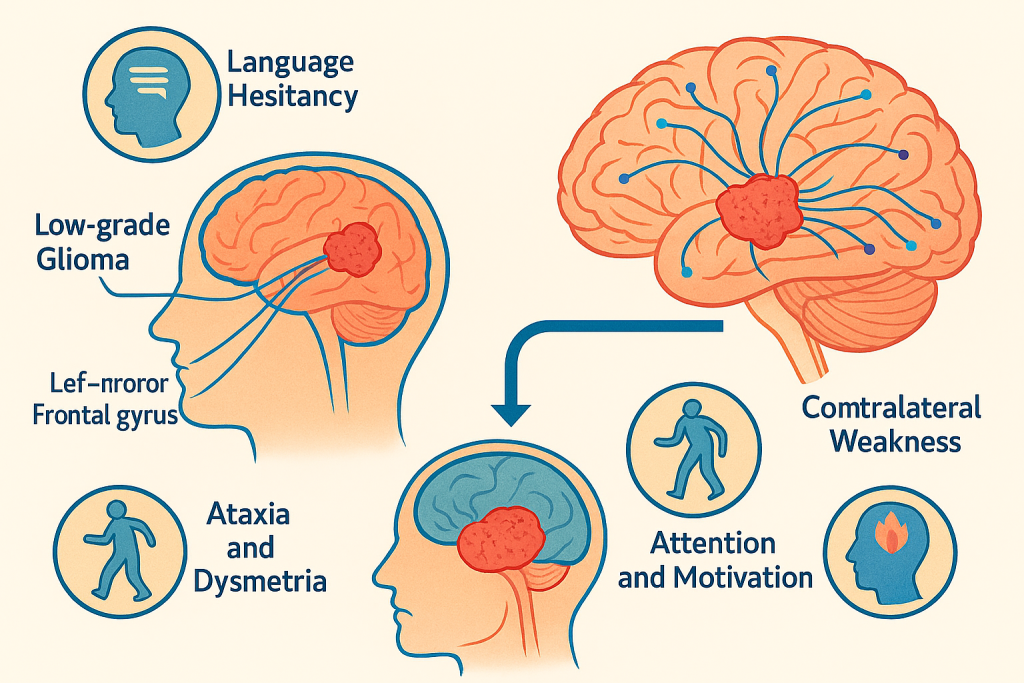
A brain tumor’s threat is not defined solely by histology or grade. Location dictates the symptom constellation and the therapeutic window. A low‑grade glioma within the left inferior frontal gyrus may precipitate subtle language hesitancy yet sit astride critical speech pathways, while a high‑grade glioblastoma in non‑eloquent frontal white matter may be resectable with comparatively lower functional risk. Motor strip lesions can cause contralateral weakness, cerebellar tumors provoke ataxia and dysmetria, and deep thalamic or basal ganglia involvement may disrupt attention and motivation. Treatment specialization thus begins with functional cartography: mapping where tumor biology intersects with the patient’s lived neurology. The clinical objective is maximal safe resection, which reduces tumor burden, improves the effectiveness of adjuvant therapy, and, in many tumor types, correlates with overall and progression‑free survival, all without sacrificing crucial neurological functions. When the lesion abuts or interdigitates with eloquent networks, the strategy shifts from purely anatomical removal to connectome‑guided preservation.
CLRD’s Diagnostic Capability: From Multi‑Modal Imaging to Molecular Stratification
The CLRD diagnostic pathway integrates structural, functional, and molecular dimensions to inform a bespoke plan of care. High‑resolution MRI with diffusion tensor imaging delineates white‑matter tracts such as the corticospinal tract, arcuate fasciculus, and inferior fronto‑occipital fasciculus, guiding the trajectory for safe microsurgical corridors. Functional MRI identifies task‑activated cortical hubs for language, motor planning, and visual processing, enabling surgeons to reconcile tumor topography with patient-specific brain function. In parallel, advanced perfusion and spectroscopy sequences help differentiate tumor progression from treatment effects and highlight metabolically active regions for targeted biopsy when tissue diagnosis is required.
Molecular profiling completes the picture. Characterization of IDH mutation, 1p/19q codeletion, MGMT promoter methylation, and other actionable markers in gliomas, as well as receptor status in metastases and specific mutations in pediatric tumors, frames prognosis and tailors adjuvant therapy. CLRD’s competency in biomarker science, developed through extensive work on circulating nucleic acids and immuno‑molecular signatures, supports non‑invasive monitoring strategies. This experience with cell‑free DNA, mitochondrial DNA dynamics, and inflammatory signaling is leveraged to design longitudinal assays for treatment response and minimal residual disease, reducing reliance on serial invasive procedures and enabling earlier therapeutic pivots when biology changes course. The net effect is a preoperative dossier that fuses imaging‑based connectivity with biological intent, setting the stage for precision intervention.
Surgical Specialization: Awake Mapping, Intraoperative Guidance, and Margin Management
Surgery remains the cornerstone of care for most surgically approachable brain tumors. CLRD’s neurosurgical partners deploy an awake–asleep–awake anesthesia paradigm for lesions encroaching on language, motor, or higher‑order networks. During awake phases, real‑time cortical and subcortical stimulation mapping elicits motor responses and language tasks, allowing the surgical team to draw a dynamic functional boundary while resecting tumor tissue. This approach is complemented by intraoperative neuronavigation that fuses preoperative tractography with surface landmarks and integrates intraoperative ultrasound or MRI to correct for brain shift, maintaining navigational accuracy as resection proceeds.
Margin management is not simply a question of removing what looks abnormal; it is a disciplined process of identifying infiltrative fronts while respecting functionally critical fibers. Intraoperative neurophysiological monitoring of motor evoked potentials and somatosensory potentials provides continuous feedback about the integrity of pathways. For extra‑axial tumors such as meningiomas, dural margins and hyperostotic bone are addressed to reduce recurrence risk, while skull‑base approaches prioritize cranial nerve preservation with endoscopic assistance where applicable. For pituitary tumors, expanded endonasal endoscopic surgery improves visualization of parasellar anatomy, facilitating more complete resection with fewer nasal and neurological complications. Throughout, the guiding principle is maximal safe resection, continuously negotiated against real‑time functional data.
When Resection is Limited: Adjuvant Radiotherapy, Chemotherapy, and Targeted Agents
Not all tumors can be wholly removed without unacceptable risk. In such cases, a sophisticated adjuvant schema is engaged. Fractionated external‑beam radiotherapy and stereotactic radiosurgery offer conformal dosing that focuses cytotoxicity on tumor residua while sparing surrounding parenchyma. Chemoradiation regimens, such as temozolomide for high‑grade gliomas, are tailored to MGMT methylation status to optimize benefit. For primary CNS lymphomas or metastases, systemic therapies are selected based on tumor biology and blood–brain barrier penetration. In parallel, targeted and immune‑modulating agents are considered where evidence supports their use.
CLRD’s translational strengths contribute decisively at this juncture. Experience with nanotechnology‑enabled delivery systems and bi‑metallic nanoparticle platforms informs strategies that are designed to ferry therapeutics across the blood–brain barrier, improve intratumoral drug concentration, and reverse resistance mechanisms at the cellular level. This technology focus, originally matured in oncology and hepatology applications, is repurposed to neuro‑oncology with careful attention to neurotoxicity thresholds and biodistribution in brain tissue. The goal is to make adjuvant therapy not just an add‑on but an integrated and potent second act in the treatment narrative.
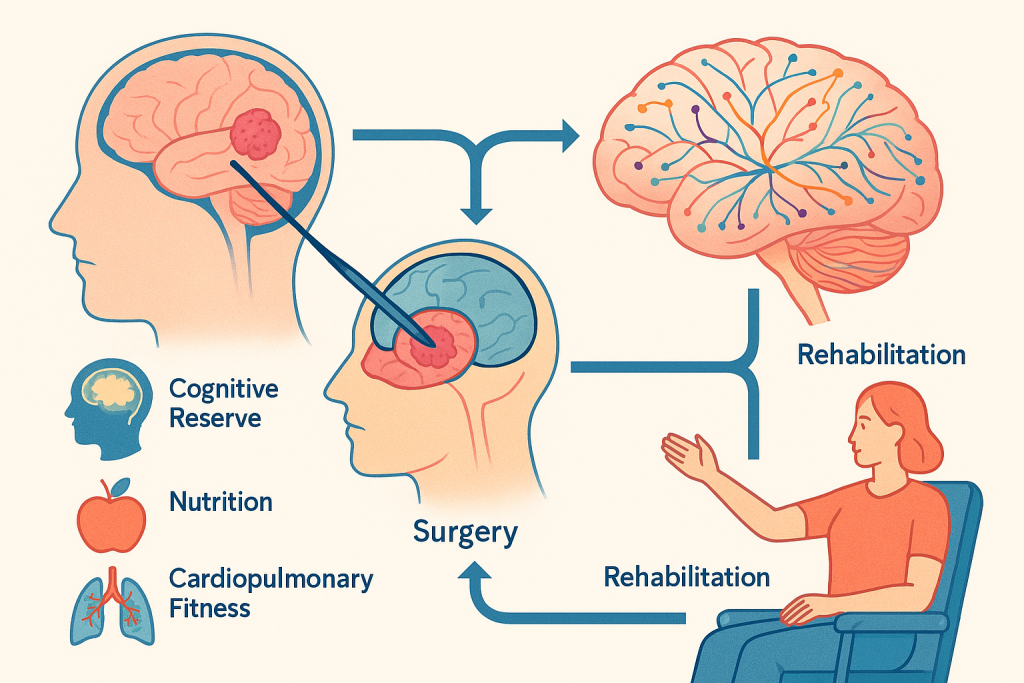
Regeneration and Recovery: CLRD’s Post‑Surgical Neuro‑Restorative Playbook
Excellence in tumor control must be matched by excellence in functional recovery. CLRD’s heritage in stem cells, organ bioengineering, and scaffold‑guided tissue repair translates into a neuro‑restorative program that begins before the first incision and extends through survivorship. Prehabilitation optimizes cognitive reserve, nutrition, and cardiorespiratory fitness to improve surgical tolerance and expedite recovery. After surgery or radiosurgery, individualized rehabilitation targets the specific neural networks displaced or partially injured by the tumor and its removal.
Building on CLRD’s foundational work in neural precursor cell biology and decellularized scaffold engineering, the center is developing adjunctive strategies for structural and synaptic support in resected cavities. While the standard of care remains evidence‑based neurorehabilitation and medical management, ongoing clinical integration efforts include the use of biocompatible, MRI‑visible materials to maintain cavity architecture for safe radiation planning and to encourage glial scar modulation. In parallel, pharmacologic neuroprotection, guided by insights into heat‑shock proteins, ABC transporters, and stress response pathways, is deployed to stabilize susceptible neural circuits during the critical postoperative window. The overarching thesis is that tumor surgery should trigger a cascade of regeneration cues, not merely conclude with wound closure.
Multidisciplinary Tumor Boards and Pathways of Care
Treatment specialization is a team sport. CLRD convenes multidisciplinary tumor boards where neurosurgeons, neuroradiologists, neuropathologists, radiation oncologists, medical oncologists, neuropsychologists, rehabilitation specialists, and clinical molecular scientists co‑author the care plan. Imaging is reviewed alongside tractography overlays and functional maps, histopathology is integrated with molecular signatures, and patient‑reported outcomes are factored into decision‑making. These boards enforce evidence‑aligned standards while granting latitude for patient preference and clinical nuance, ensuring that innovation is disciplined by safety and scientific plausibility.
Pathways are codified for common tumor types. For diffuse low‑grade gliomas, early resection with connectome‑guided techniques is paired with watchful molecular surveillance and, when indicated, adjuvant therapy calibrated to risk. For glioblastoma, tri‑modality therapy is coordinated from the outset, incorporating surgical cytoreduction, concurrent chemoradiation, and maintenance therapy with rigorous imaging follow‑up. Skull‑base meningiomas are addressed with endoscopic or open approaches depending on extent and relationship to cranial nerves and vascular structures, with adjuvant radiosurgery for residual plaque or cavernous sinus involvement. Pituitary adenomas are stratified by hormonal activity and optic apparatus compromise, aligning transnasal endoscopic surgery with endocrinologic co‑management. Metastatic disease is navigated with a balance of systemic control, focal radiosurgery, and surgery for mass effect or diagnostic clarification, mindful of the patient’s systemic disease trajectory.
Precision Supportive Care: Seizures, Edema, Endocrine Health, and Cognition
Seizure management is tailored with antiepileptic drugs selected for minimal cognitive and hematologic side effects and compatibility with oncologic regimens. Peritumoral edema is addressed judiciously with corticosteroids, carefully tapered to avoid immunosuppression during chemotherapy or heightened infection risk. For pituitary and hypothalamic region tumors, endocrine evaluation and replacement are proactive to prevent adrenal crises, thyroid imbalance, and gonadal axis disruption. Cognitive health is actively measured and supported; neuropsychological testing guides cognitive rehabilitation, and risk factors for fatigue, depression, and anxiety are preemptively managed. The emphasis is not only on survival but on preserved identity and quality of life.
CLRD’s Distinctive Value: Translational Engineering Applied to Neuro‑Oncology
What differentiates CLRD in brain tumor care is the union of meticulous surgical and oncologic practice with regenerative engineering and precision biomarker science. Decades of work on cellular therapies and organ‑scale scaffolds translate into an unusual competence in tissue interfaces and healing biology, which is directly relevant to brain surgery’s delicate balance between removal and repair. Expertise with nanoparticle‑based drug delivery and radio‑sensitization strategies enhances the effectiveness of adjuvant treatments in a compartment where the blood–brain barrier has historically hamstrung pharmacology. Proficiency in circulating biomarkers and mitochondrial/nuclear DNA analytics opens promising avenues for earlier detection of recurrence and for titrating therapy intensity to real‑time biology rather than static schedules.
These capabilities do not replace the standards of care; they augment them. Awake mapping still safeguards eloquent function, conformal radiotherapy still sterilizes residual cells, and proven chemotherapies remain backbone agents. CLRD’s contribution is to densify each link in that chain—richer diagnostics inform better surgery, smarter delivery platforms empower adjuvant therapy, and structured regeneration science accelerates recovery.
Patient Journey at CLRD: From First Symptom to Sustained Survivorship
Care begins with expedited evaluation, typically prompted by new neurological symptoms or an incidental imaging finding. Within a compressed timeframe, the patient undergoes high‑definition MRI with tractography and functional mapping where indicated, targeted laboratory workup, and a molecular strategy session to define tissue needs. If surgery is appropriate, prehabilitation and counseling prepare the patient for awake mapping and postoperative milestones. The operation itself is a choreography of mapping, resection, and verification, navigation updated against intraoperative imaging, physiology monitored continuously, and functional tasks elicited as needed.
Postoperatively, imaging confirms the extent of resection and establishes a new baseline. Adjuvant therapy is then sequenced deliberately: consultations in radiation and medical oncology finalize a plan that respects molecular drivers and patient priorities. Throughout, rehabilitation is active and adaptive, with speech, occupational, and physical therapy engaging specific deficits and measuring gains. Survivorship is structured around periodic imaging, circulating biomarker monitoring where appropriate, cognitive check‑ins, endocrinologic assessment for relevant tumor types, and return‑to‑life milestones including work, driving, and recreation. Psychological support and peer mentorship are integrated to contextualize the journey and mitigate isolation.
Safety, Ethics, and Continuous Quality
Innovation in the brain demands humility. CLRD maintains guardrails through rigorous consent processes, staged introductions of novel adjuncts only when they align with established safety profiles, and outcome tracking that feeds back into practice. Case reviews, morbidity and mortality conferences, and registry participation ensure that lessons are codified and disseminated. Patients are partners in this process, fully briefed not only on treatment choices but on the rationale, alternatives, and contingencies.
In Summary: Precision Against the Tumor, Preservation for the Person
Brain tumors can fracture the continuity of self by threatening the neural networks that underwrite movement and meaning. Treatment specialization at CLRD is constructed to resist that fracture: diagnostics that reveal both structure and function, surgery that negotiates margins with live maps, adjuvant therapy empowered by intelligent delivery, and recovery strategies that actively promote neural resilience. The result is a care model that aims not only to excise disease but to conserve capability—anchored in the best of contemporary neuro‑oncology and elevated by CLRD’s translational strengths in regeneration, bioengineering, and precision molecular medicine.
